Inhaled and Intranasal Products: Contract Service Providers Market (Focus on Drugs and Drug Delivery Systems), 2020 – 2030 | Market Size | Industry Analysis | 2030

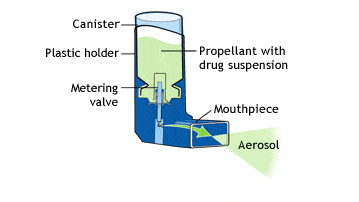
PROCESS VALIDATION AND REGULATORY REQUIREMENTS OF METERED-DOSE INHALERS: AN OVERVIEW | Semantic Scholar

How to match the optimal currently available inhaler device to an individual child with asthma or recurrent wheeze | npj Primary Care Respiratory Medicine

PDF) Raman spectroscopy for the process analysis of the manufacturing of a suspension metered dose inhaler | Luis De la Cruz - Academia.edu

PDF) Influence of Formulation Factors on the Aerosol Performance of Suspension and Solution Metered Dose Inhalers: A Systematic Approach

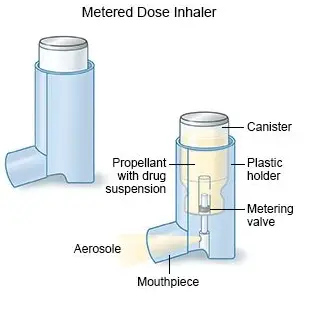
![PDF] FORMULATION AND QUALITY CONTROL OF METERED DOSE INHALER : A REVIEW | Semantic Scholar PDF] FORMULATION AND QUALITY CONTROL OF METERED DOSE INHALER : A REVIEW | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d66c8646b90ac455a9b5dbdd41180ff6aa82337e/2-Figure2-1.png)